Abstract
The pathogenesis of idiopathic aplastic anemia (IAA) involves a human leukocyte antigen (HLA)-restricted T-cell autoreactivity against unknown antigens preferentially distributed on early hematopoietic stem and progenitor cells (HSPCs). Genetically acquired GPI-anchor and HLA deficiency have been both linked to clonal immune evasion from T-cell pressure. We hypothesized that, in analogy to anti-tumor adaptive immune evasion, pathophysiology of immune escape in IAA originates together with a broader dysfunction of antigen presentation/processing machinery and immune regulatory proteins, beyond HLA molecules, as an effect of immune pressure under T-cell attack. This initial immune reaction would produce up-modulation of these pathways, ultimately promoting the acquisition of mutations and expansion of immune resistant clones.
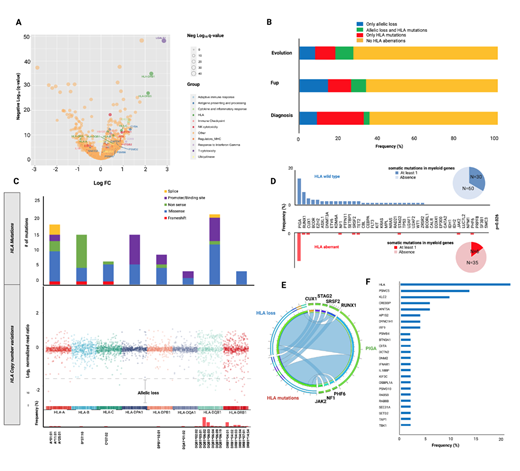
To test this hypothesis, we first performed single-cell RNAseq analysis in HSPCs in IAA patients at disease manifestation, 1 which showed signatures of dysfunction of antigen presentation machinery, with up-regulation of most of the HLA molecules, proteasome subunits and endoplasmic reticulum related organelle transporters. Strikingly, DRB1 was among the top 3 genes upregulated in IAA patients compared to controls (q-values 1.23E-35; Fig.1A), underscoring the etiological impact that antigen presentation via this locus has in the initiation of autoimmune process. Mild upregulation was also seen in DQB1 and B loci (q-values 4.7E-07 and 2.1E-10, respectively).
We then studied molecular escape mechanisms by genotyping 204 IAA and PNH patients, with either a targeted or whole genome sequencing (WGS) platform. By application of a newly in-house developed bioinformatic pipeline, we detected somatic aberrations in HLA region involving both class I and II alleles in 36% of IAA patients including point mutations, frameshift insertions or deletions and copy number variations inducing allelic loss. B*14:02 and A*02:01 emerged as the most commonly mutated class I alleles with a few hotspot mutations identified, particularly in exon 1 (c.19C>T, p.R7X, Fig.1B,C), confirming previous reports. 2,3 In class II, DQB1 and DPA1 loci were frequently targeted by fine mutational events, while more complex allelic loss phenomena interested prevalently DRB1 and DQB1 loci. Those aberrations were identified at diagnosis (35%), during disease follow-up (33%) or at the time of malignant evolution (27%), with higher clonal size in specimens collected during the course of the disease (median VAF 3% [2-27%] at diagnosis, 8% [2-98%] at follow-up, and 2.2% [2.0-6.1%] at evolution). Of 41 patients with at least one HLA aberration and characterized with an extended genotypic study, only 6 harbored also >1 somatic myeloid mutation (14%), versus 30/90 (33%) not affected by somatic hits in HLA (p=.026; Fig.1D). HLA aberrant cases also showed lower number of somatic myeloid mutations (OR=0.44; p=.0262) with driver hits rarely present (Fig.1E). In terms of PIGA mutations, an increased PIGA mosaicism was observed in the HLA mutant group, underlying that both processes have similar pathophysiologic origin as a product of the immune selection pressure (OR: 1.55 [95%CI 1.1-4.2], p=.0201). We then investigated, through WGS of 53 patients, the presence of somatic mutations in other immune genes which could be triggered by immune pressure. Hence, in 47% of the cases we were able to find pathogenic or likely pathogenic hits in genes encompassing proteasome complex, vesicle trafficking, transactivators and interferon regulatory factors, including CREBBP, TAP1, CIITA, PSMC5, PSMB4 and IRF9 (Fig.1F), whose pathogenicity was computationally assessed through recently implemented somatic classifiers. 4 Those hits were not mutually exclusive neither with HLA nor with PIGA mutations, however their VAF was significantly lower compared to concurrent HLA and PIGA lesions, underscoring their lower driver potential within the immune escape environment compared to PNH and HLA-lacking clones.
Altogether our results describe the diversity of molecular and immune events taking place in IAA and PNH. Our study suggests that following initial immune insult, clonal architecture of residual hematopoiesis can be dominated by multiple modes of immune escape, agonistically participating to a mechanism of "adaptive" clonal recovery, likely in opposition to the "maladaptive" malignant progression.
Maciejewski: Alexion: Consultancy; Regeneron: Consultancy; Novartis: Consultancy; Bristol Myers Squibb/Celgene: Consultancy.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal